Okay, feed is kind of a misnomer, but it's close. Malaria parasites that we work with in the lab are cultured in human blood, and they infect the red blood cells within the blood. They do harvest some of the stuff that's in the red blood cell, but they also get nutrients from the media we use, which is formulated to be similar to human plasma.
So--a red blood cell is the main vehicle for exchanging oxygen and carbon dioxide in your body, and they are very, very good at it. This is mostly because they are stuffed to the brim full of a protein called hemoglobin, which in turn has four heme molecules inside it, each with an iron held in place in the center (the iron, seen below in yellow, is what helps the oxygen bind to the hemoglobin, believe it or not!).
from Sigma Aldrich
Now, when a parasite invades a red blood cell, it has a problem. It's new home is full of other stuff...hemoglobin! And the parasite needs space to grow. So, it begins to break down the hemoglobin proteins, but the heme groups are a different story. In cells, free iron is actually toxic, so releasing it would be a bad thing.
Instead, the parasite has this marvelous trick; it takes the heme group and turns it into a crystal called hemozoin to make sure the iron is sequestered away:
from Wikipedia
In turn, scientists like me have made use of the parasite's iron sequestration to help us with research...our own neat trick! Iron inside hemozoin is diamagnetic, but when the parasite crystallizes it into hemozoin, the iron is actually paramagnetic (citation here).
While you may not understand the technical descriptions of paramagnetism versus diamagnetism, you know that there are some things in this world that are diamagnetic...ceramic coffee cups, paper, plastic bottles. They don't stick to magnetized surfaces. However, paramagnetic things like refrigerator magnets do in fact stick to magnetized surfaces.
This means that the parasite-infected cell is magnetic...it will stick to a magnet! And science has designed a column that allow the infected cells to stick to magnetic beads inside it, and let all the other stuff flow through. This allows us to have a solution of concentrated infected cells for experiments...a process we call enrichment.
Step 1: Check the parasitemia.
Big word, easy concept. I need to know what percentage of red blood cells have a parasite in them and make sure it's within the right range. I smear blood from our cultures onto a glass slide, fix it with methanol, and then dye it with Giemsa.
Giemsa gets...everywhere. Literally. I spilled some on my lab coat a year ago, and it's going to be there forever. It's the dye equivalent of what would survive the apocalypse.
I use a 100X objective to look at the cells under the microscope. The dots of oil are required for the microscope to look at something that small (a red blood cell is about 10 µm, or micrometers, meaning about 20 of them in a row would be the same size as the diameter of a human hair).
The Niles lab is old school...this is a counting apparatus to allow for counting the parasites of different ages while we look under the microscope. For some experiments, knowing the age of the population is actually really important.
So, now that I know that the parasitemia is in the right range and the parasites are the right age, I can go forward with the experiment!
[an aside...this is the part that's actually really tricky about culturing. Having the parasites be the right age and having the right amount inside a flask is much harder than it looks, and it takes a lot of time to ensure that your cultures are happy, and ready when you need them. Hence why I spend at least an hour on just basic culturing every day, including Saturday and Sunday. It's definitely a skill, with about 5% uncertainty mixed in, much to my chagrin. Sometimes all your cultures just crash, for no discernible reason. Obnoxious.]
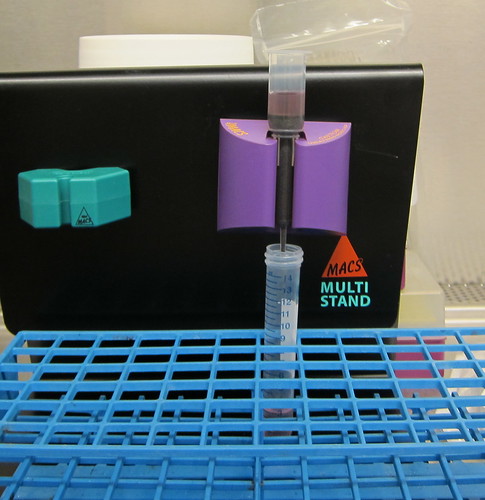
Step 2: Set up the column.
So, now that I know that the parasitemia is in the right range and the parasites are the right age, I can go forward with the experiment!
[an aside...this is the part that's actually really tricky about culturing. Having the parasites be the right age and having the right amount inside a flask is much harder than it looks, and it takes a lot of time to ensure that your cultures are happy, and ready when you need them. Hence why I spend at least an hour on just basic culturing every day, including Saturday and Sunday. It's definitely a skill, with about 5% uncertainty mixed in, much to my chagrin. Sometimes all your cultures just crash, for no discernible reason. Obnoxious.]
Step 2: Set up the column.
--the black MACS Multistand is magnetic and has two holders for columns that are teal and purple. --the thing in the purple holder is a MACS column
--the black stuff inside the MACS column is magnetic beads.
--the red stuff on top in the MACS column is the media we use, and we put in on the column to wash it and make it more hospitable for cells.
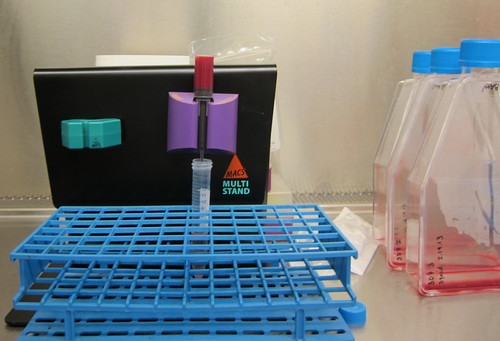
Step 3: Prepare cells.
--the black stuff inside the MACS column is magnetic beads.
--the red stuff on top in the MACS column is the media we use, and we put in on the column to wash it and make it more hospitable for cells.
Step 3: Prepare cells.
A peek into the incubator -- you can see some plates in the back, and the red capped things that look like they have flowers on them are 10 mL flasks. This is the normal size for regular maintenance. I'm using rather large blue-capped ones for a 30 mL culture (you can see them off to the side in the next picture). Since red blood cells are heavier than the media, they are on the bottom of the flask, making it easy to remove the media. At that point, I can bring them up in a volume that fits on the column (in this case, the bright red that you see in the picture below is 6 mL).
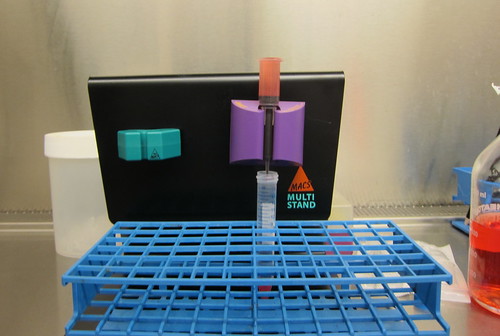
Step 4: Apply infected red blood cells to the MACS column.
Step 4: Apply infected red blood cells to the MACS column.
It takes about 30 minutes for this viscous blood and media mixture to drip through the column.
Almost there!
Step 5: Wash column.
Step 5: Wash column.
Because I'm using such a high number of cells, it's a good idea to send a bit more liquid through to make sure that the things that are stuck to the column are the magnetic infected red blood cells I want.
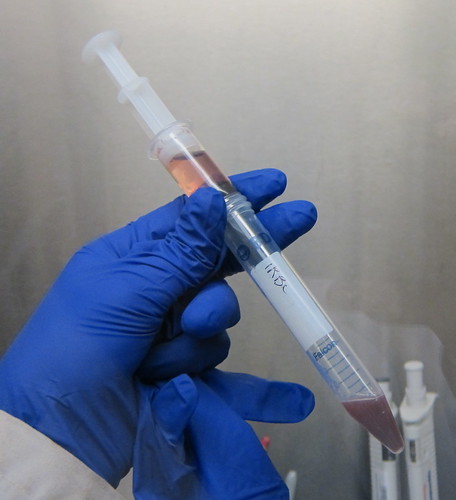
Step 6: Elute!
Step 6: Elute!
Look at this contraption! To speed up the process of getting the infected cells from off the column, I first remove the column from the magnet, and then use a syringe to flow liquid through that will take the infected cells with it!
Step 7: Spin down, and you're done.
Step 7: Spin down, and you're done.
At this point, I put this conical tube into a centrifuge, and it pellets the heavy infected cells to the bottom of the tube.
Congratulations, you have parasite-infected cells ready for experiments!
(writing this protocol up reminded me that each of these pellets represents about 15 hours of work...yikes)
Congratulations, you have parasite-infected cells ready for experiments!
(writing this protocol up reminded me that each of these pellets represents about 15 hours of work...yikes)







No comments:
Post a Comment